
Wu, Chih-Hang (吳志航)
Associate Research Fellow
- Ph.D., Biology, University of East Anglia, UK
- Plant Immunity; Molecular Plant-microbe Interactions
- wuchh@gate.sinica.edu.tw
- wuchh@as.edu.tw
- +886-2-2787-1142 (Lab: )
- +886-2-2787-1141 (Office: R420)
- Lab Website
- Academia Sinica Archive
- ORCID
- Web of Science (WOS)
- Google Scholar
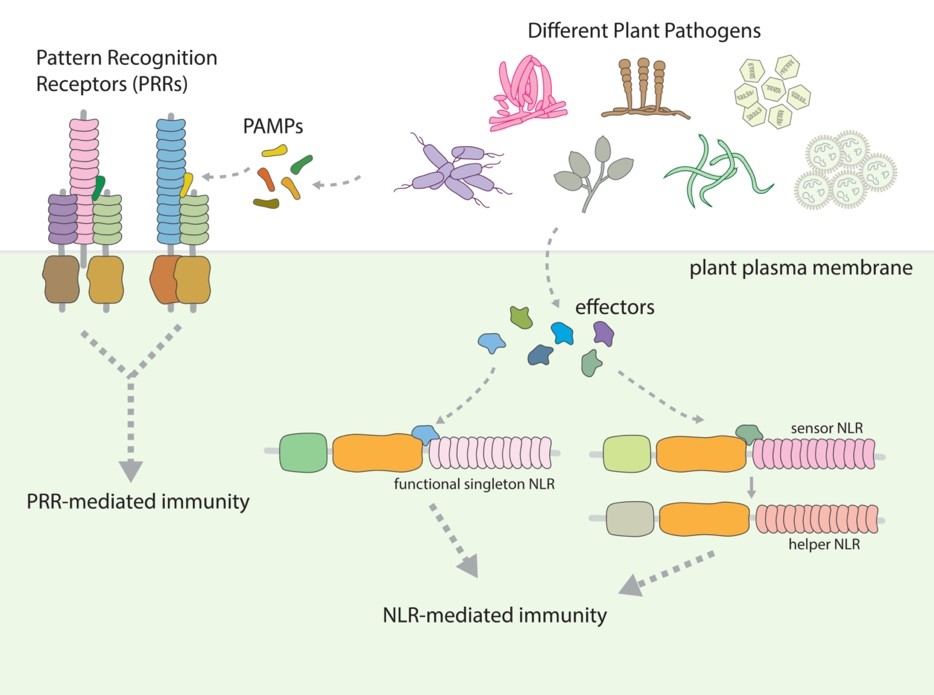
The plant innate immune system
Plants are continuously exposed to diverse microorganisms in the environment, many of which can invade plants and cause diseases. Some of these plant pathogenic microorganisms infect economically important crops, leading to huge yield loss in agriculture. To fend off the invading pathogens, plants have developed an immune system to detect pathogens and restrict pathogen growth. This includes using the cell surface PRRs (Pattern Recognition Receptors) and intracellular NLRs (Nucleotide-binding domain Leucine-rich repeat Receptors) to recognize pathogen molecules and then activate immune signaling. Many of these immune receptors function as disease resistance (R) proteins that protect plants from pathogen invasion and are very useful in agriculture.
PRRs localize at the plasma membrane and detect extracellular PAMPs (Pathogen-Associated Molecular Patterns), whereas NLRs are intracellular proteins that detect the effector proteins secreted from pathogens. Upon the recognition of pathogen molecules, these immune receptors activate downstream signaling cascades, leading to PRR-mediated immunity and NLR-mediated immunity. Some NLRs work as functional singletons that detect pathogens and activate immune response on their own; however, some NLRs function together, in which one of them is a sensor NLR that detect pathogen and the other one is a helper NLR that is essential for immune signaling.
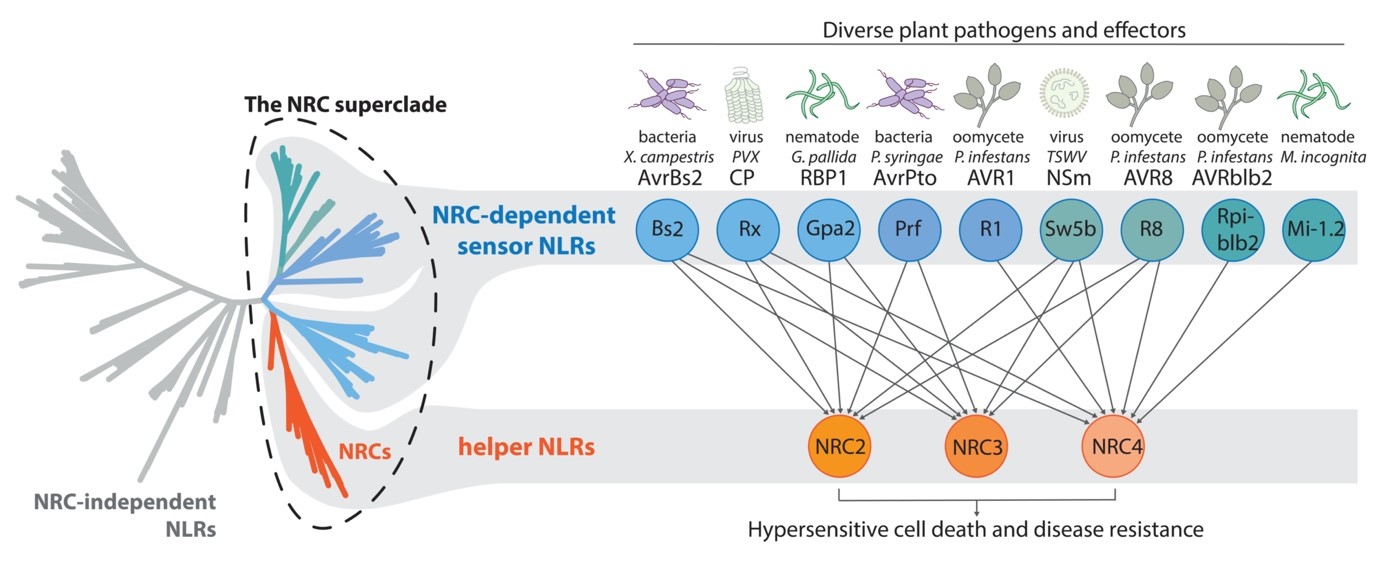
The NRC network of solanaceous plants
Recent discoveries showed that NLRs can work as in functional singletons, pairs and networks. In the solanaceous plants, the NRC network that confers resistance to various pathogens is composed of several sensor NLRs that detect different pathogen proteins, and three major helper NLRs (NRC2, NRC3 and NRC4) that are functionally redundant but display distinct specificities toward different sensor NLRs. Furthermore, the NRC family and NRC-dependent sensor NLRs are phylogenetically clustered into a well-supported superclade. These results reveal a complex genetic network beyond the ‘gene-for-gene’ hypothesis, and link immune signaling to the history of NLR gene evolution.
Our research center on the evolutionary and functional dynamics of the plant immunity. We hope to address the following three questions about the NRC network:
- How do helper-sensor NLR proteins function together?
- How does the NRC network specialize in different plant tissues?
- How did the NRC network evolve in different plant lineages?
- Selvaraj M, Toghani AA, Pai H, Sugihara Y, Kourelis J, Yuen ELH, Ibrahim T, Zhao H, Xie R, Maqbool A, De la Concepcion JC, Banfield MJ, Derevnina L, Petre B, Lawson DM, Bozkurt TO, Wu CH, Kamoun S and Contreras MP. 2024. Activation of plant immunity through conversion of a helper NLR homodimer into a resistosome. PLOS Biologyhttps://doi.org/10.1371/journal.pbio.3002868
- Huang CY#, Huang YS#, Sugihara Y, Wang HY, Huang LT, Lopez-Agudelo JC, Chen YF, Lin KY, Chiang BJ,Toghani AA, Kourelis J, Wang CH, Derevnina L, Wu CH*. 2024. Subfunctionalization of NRC3 altered the genetic structure of the NicotianaNRC network. PLOS Genetics https://doi.org/10.1371/journal.pgen.1011402 (#Equal contribution, *Corresponding author)
- Chiang BJ#, Lin KY#, Chen YF, Huang CY, Goh FJ, Huang LT, Chen LH, Wu CH*. 2024. Development of a tightly regulated copper-inducible transient gene expression system in Nicotiana benthamianaincorporating suicide exon and Cre recombinase. New Phytologist https://doi.org/10.1111/nph.20021 (#Equal contribution, *Corresponding author)
- Goh FJ, Huang CY, Derevnina L, Wu CH*. 2024. NRC immune receptor networks show diversified hierarchical genetic architecture across plant lineages. The Plant Cell https://doi.org/10.1093/plcell/koae179 (*Corresponding author)
- Sakai T, Contreras MP, Martinez-Anaya C, Lüdke D, Kamoun S*, Wu CH*, Adachi H*. 2024. The NRC0 gene cluster of sensor and helper NLR immune receptors is functionally conserved across asterid plants. The Plant Cell https://doi.org/10.1093/plcell/koae154 (*Corresponding author)
- Chen JY, Sang H, Chilvers MI, Wu CH, Chang HX. 2024. Characterization of soybean chitinase genes induced by Rhizobacteria involved in the defense against Fusarium oxysporum. Plant Sci.doi: 10.3389/fpls.2024.1341181
- Wu CH and Derevnina L. 2023. The battle within: How pathogen effectors suppress NLR-mediated immunity. Current Opinion in Plant Biology org/10.1016/j.pbi.2023.102396
- Sheikh AH, Zacharia I, Pardal1 AJ, Dominguez-Ferreras A, Sueldo DJ, Kim JG, Balmuth A, Gutierrez JR, Conlan BF, Ullah N, Nippe OM, Girija AM, Wu CH, Sessa G, Jones AME, Grant MR, Gifford ML, Mudgett MB, Rathjen JP and Ntoukakis V. 2023. Dynamic changes of the Prf/Pto tomato resistance complex following effector recognition. Nature Communications 14: 2568. org/10.1038/s41467-023-38103-6
- Contreras MP, Pai Hsuan, Selvaraj M, Toghani AA, Lawson DM, Tumtas Y, Duggan C, Yuen ELH, Stevenson CEM, Harant A, Wu CH, Bozkurt TO, Kamoun S, Derevnina L. Resurrection of plant disease resistance proteins via helper NLR bioengineering. 2023. Science Advances 9, eadg3861. DOI: 10.1126/sciadv.adg3861
- Oh S, Kim S, Park HJ, Kim MS, Seo MK, Wu CH, Lee HA, Kim HS, Kamoun S, Choi D. 2023. Nucleotide-binding leucine-rich repeat network underlies nonhost resistance of pepper against the Irish potato famine pathogen Phytophthora infestans. Plant Biotechnology Journal org/10.1111/pbi.14039
- Adachi H, Sakai T, Harant A, Duggan C, Bozkurt TO, Wu CH#, Kamoun S#. 2023. An atypical NLR protein modulates the NRC immune receptor network. PLOS Genetics 19(1): e1010500. org/10.1371/journal.pgen.1010500(#Corresponding author)
- Ahn HK, Lin X, Olave-Achury AC, Derevnina L, Contreras MP, Kourelis J, Wu CH, Kamoun S, Jones JDG. 2023. Effector-dependent activation and oligomerization of plant NRC class helper NLRs by sensor NLR immune receptors Rpi-amr3 and Rpi-amr1. EMBO J e111484. org/10.15252/embj.2022111484
- Contreras MP, Pai H, Tumtas Y, Duggan C, Yuen ELH, Cruces AV, Kourelis J, Ahn HK, Lee KT, Wu CH, Bozkurt TO, Derevnina L, Kamoun S. 2023. Sensor NLR immune proteins activate oligomerization of their NRC helper. EMBO J doi.org/10.15252/embj.2022111519
- Kourelis J, Contreras MP, Harant A, Pai H, Lüdke D, Adachi H, Derevnina L, Wu CH#, Kamoun S#. 2022. The helper NLR immune protein NRC3 mediates the hypersensitive cell death caused by the cell-surface receptor Cf-4. PLOS Genetics 18(9): e1010414. org/10.1371/journal.pgen.1010414 (#Corresponding author)
- Lin X, Olave-Achury A, Heal R, Witek K, Karki HS, Song T, Wu CH, Adachi H, Kamoun S, Vleeshouwers VGAA, Jones JDG. 2022. A potato late blight resistance gene protects against multiple Phytophthora species by recognizing a broadly conserved RXLR-WY effector. Molecular Plant 15(9): 1457-1469. org/10.1016/j.molp.2022.07.012
- Derevnina L, Contreras MP, Adachi H, Upson JL, Cruces AV, Xie R, Sklenar J, Menke FLH, Mugford ST, MacLean D, Ma W, Hogenhout S, Goverse A, Maqbool A, Wu CH#, and Kamoun S#. 2021. Plant pathogens convergently evolved to counteract redundant nodes of an NLR immune receptor network. PLOS Biology 19(8): e3001136. org/10.1371/journal.pbio.3001136 (#Corresponding author)
- Duggan C, Moratto E, Savage Z, Hamilton E, Adachi H, Wu CH, Leary AY, Tumtas Y, Maqbool A, Kamoun S, Bozkurt TO. 2021. Dynamic accumulation of a helper NLR at the plant-pathogen interface underpins pathogen recognition. PNAS 118:e2104997118 org/10.1073/pnas.2104997118
- Duxbury Z, Wu CH#, Ding P#. 2021. A comparative overview of the intracellular guardians of plants and animals: NLRs in innate immunity and beyond. Rev. Plant Biol 72:155-184 doi.org/10.1146/annurev-arplant-080620-104948 (#Corresponding author)
- Witek K, Lin X, Karki HS, Jupe F, Witek AI, Steuernagel B, Stam R, van Oosterhout C, Fairhead S, Heal R, Cocker JM, Barrett W, Wu CH, Adachi H, Song T, Kamoun S, Vleeshouwers VGAA, Tomlinson L, Wulff BBH, Jones JDG. 2021. A complex resistance locus in Solanum americanum recognizes a conserved Phytophthora Nature Plants 7: 198–208. doi.org/10.1038/s41477-021-00854-9
- Wu CH and Kamoun S. 2021. Tomato Prf requires NLR helpers NRC2 and NRC3 to confer resistance against the bacterial speck pathogen Pseudomonas syringae tomato. Acta Hortic. 1316, 61-66 doi.org/10.17660/ActaHortic.2021.1316.9 bioRxiv: doi.org/10.1101/595744
- Gao C, Xu H, Huang J, Sun B, Zhang F, Savage Z, Duggan C, Yan T, Wu CH, Wang Y, Vleeshouwers VGAA, Kamoun S, Bozkurt TO, Dong S. 2020. Pathogen manipulation of chloroplast function triggers a light-dependent immune recognition. PNAS 117:9613-9620 org/10.1073/pnas.2002759117
- Wu CH*, Adachi H*, De la Concepcion JC*, Castells-Graells R, Nekrasov V, Kamoun S. 2020. NRC4 gene cluster is not essential for bacterial flagellin-triggered immunity. Plant Physiology 182: 455–459. org/10.1104/pp.19.00859 (* Equal contribution)
- Frantzeskakis L, Pietro A Di, Rep M, Schirawski J, Wu CH, and Panstruga R. 2020. Rapid evolution in plant–microbe interactions–a molecular genomics perspective. New Phytologist 225 :1134-1142. org/10.1111/nph.15966

Wen-Chi Hu (胡文綺)
Ph.D. Plant Pathology and Microbiology, National Taiwan University
wenchi6276@gate.sinica.edu.tw
+886-2-27871142 (R422)

Liang-Yu Hou (侯良諭)
Ph.D. Biology, Ludwig-Maximilians-Universität München, Germany
a090716@gate.sinica.edu.tw
+886-2-27871142 (R420)

Hung-Yu Wang (汪紘宇)
National Chung-Hsing University (TIGP-MBAS)
M.S. Bioscience and Biotechnology, National Taiwan Ocean University
hank99911@gmail.com
+886-2-27871142 (R422)

Juan Carlos Lopez (洛胡安)
National Chung-Hsing University (TIGP-MBAS)
M.S. Molecular Genetics and Biotechnology, University of Seville, Spain
as0200314@gate.sinica.edu.tw
+886-2-27871142 (R420)

Sopio Tchabashvili (莎平歐)
National Chung-Hsing University (TIGP-MBAS)
M.S. Agriculture Sciences, Georgian Technical University, Georgia
tchabashvil0001@gate.sinica.edu.tw
+886-2-27871142 (R420)

Kuan-Yu Lin (林冠妤)
National Taiwan University
b08608046@ntu.edu.tw
+886-2-27871142 (R422)

Chin-Wen Chang (張槿玟)
M.S. Plant Biology, National Taiwan University
jwchang@gate.sinica.edu.tw
+886-2-27871142 (R420)

Bing-Jen Chiang (江秉真)
M.S. Plant Pathology and Microbiology, National Taiwan University
as0191504@gate.sinica.edu.tw
+886-2-27871142 (R420)

Yi-Feng Chen (陳宜豐)
M.S. Plant Pathology and Microbiology, National Taiwan University
yfchen00@gate.sinica.edu.tw
+886-2-27871142 (R420)

Wen-Rong Hsiao (蕭文榮)
M.S. Plant Pathology, National Chung-Hsing University
wrhsiao@gate.sinica.edu.tw
+886-2-27871142 (R420)
International
- 2024: EMBO Global Investigator, European Molecular Biology Organization (EMBO)
Domestic
- 2025: Young Scholars’ Creativity Award , Foundation for the Advancement of Outstanding Scholarship (FAOS)
- 2025: Career Development Award , Academia Sinica
- 2024: Shang-Fa Yang Young Scientist Award , The Shang-Fa Yang Memorial Foundation
- 2024: Outstanding Young Scholar Award, Taiwan Society of Plant Biologists
- 2022: Emerging Young Scholars (2030 Cross-Generation Young Scholars Program) , Ministry of Science and Technology
This PI participates in the following graduate programs.