[Pao-Yang Chen] EED is required for Mouse Primordial Germ Cell Differentiation in the Embryonic Gonad
POST: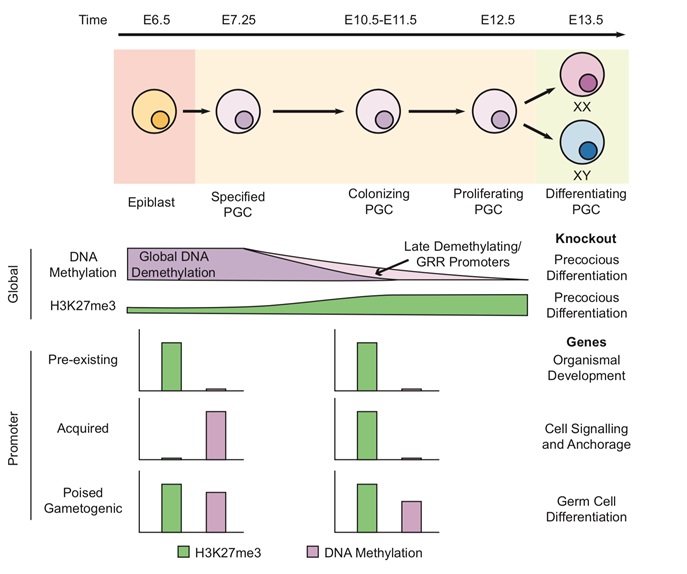
DNA methylation and H3K27me3 are required to prevent precocious PGC differentiation in response to sex-specific developmental signals during sex determination in the embryonic ovary and testis. Three types of genes regulation are identified during PGC development.
Promoters that exhibit H3K27me3 in both epiblast PGCs are involved in organismal development; Promoters begin with high levels of DNA methylation in the epiblast and then gain H3K27me3 after loss of DNA methylation are involved in cell signaling and anchorage.
Promoters with high levels of DNA methylation and H3K27me3 in epiblast in both epiblast and PGCs are related to gametogenesis.
Development of Primordial germ cells (PGCs) is required for reproduction. During PGC development in mammals, major epigenetic remodeling occurs which is hypothesized to establish an epigenetic landscape for sex-specific germ cell differentiation and gametogenesis. In order to address the role of Embryonic Ectoderm Development (EED) and Histone 3 lysine 27 trimethylation (H3K27me3) in this process, we created a conditional deletion in EED and show that EED is essential for regulating the timing of sex-specific PGC differentiation in both ovaries and testes, as well as X chromosome dosage decompensation in testes. Integrating chromatin and whole genome bisulfite sequencing of epiblast and PGCs, we identified a poised repressive signature of H3K27me3/DNA methylation which we propose is established in the epiblast where EED and DNMT1 interact. Thus, EED joins DNMT1 in regulating the timing of sex-specific PGC differentiation during the critical window when the gonadal niche cells specialize into an ovary or testis.
Link:https://www.sciencedirect.com/science/article/pii/S1534580722003690