[Paul Verslues] Protein Phosphatases: the flip side of phosphosignalling
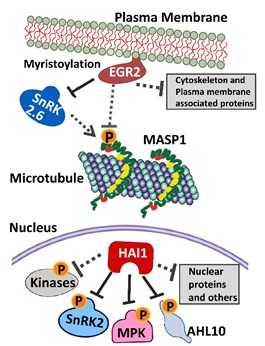
POST:Protein phosphorylation is one of the most widespread post-translational modifications which affect protein function. Phosphorylation is important in many types of signaling in part because it is dynamic. Phosphorylation by protein kinases can be readily reversed by protein phosphatases. Improved analytical techniques have led to greater appreciation of the vastness of the plant phosphoproteome. In the model plant Arabidopsis, there may be as many as 35,000 protein phosphorylation sites on more than 6000 phosphoproteins. In a recent review (Bhaskara et al., 2019) considered the question of why plants have more then 1000 kinases for protein phosphorylation yet, the flip side of protein dephosphorylation is managed by only about 150 phosphatases. Of these protein phosphatases, by far the largest group is the type 2C protein phosphatases (PP2Cs), which have greatly expanded numbers in plants compared to other eukaryotes. We also described new data on function of individual PP2Cs such as the EGR PP2Cs, which the Verslues laboratory and others have found to be new players in abiotic stress signaling.