[Chih-Hang Wu] Uncovering the key structural mechanism of plant resistosomes
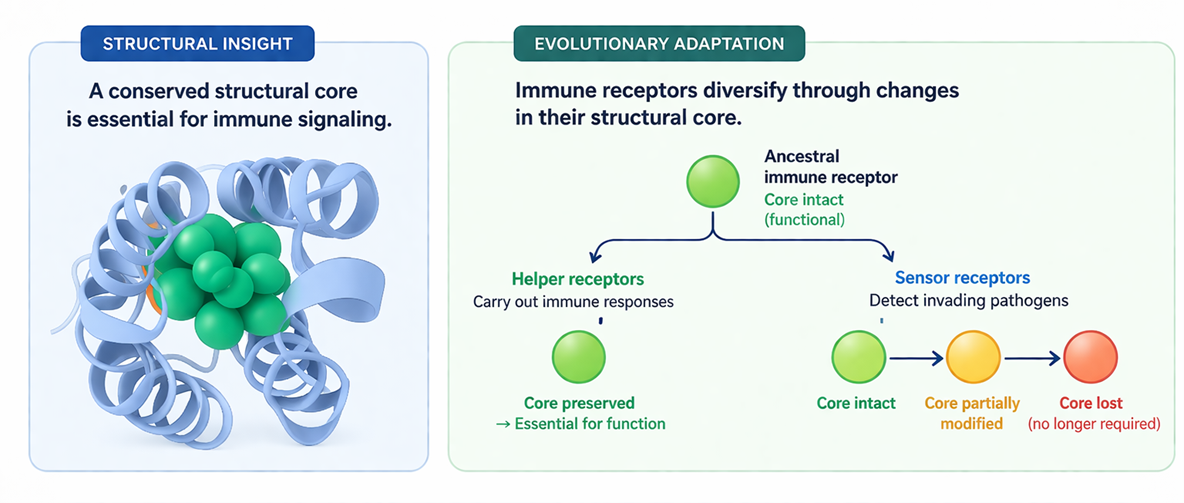
POST:In response to pathogen attack, plants rely on NLR (nucleotide-binding leucine-rich repeat) receptors to activate immune responses. The NRC (NLR required for cell death) family acts as “helper NLRs” that enable sensor NLRs to trigger hypersensitive cell death, yet their molecular mode of action remains unclear.
In this study, we examined the N-terminal coiled-coil (CC) domain of NRC proteins. Using site-directed mutagenesis and transient expression in Nicotiana benthamiana, we identified seven residues essential for NRC4-mediated cell death, four of which form a stable “hydrophobic core.” This core is critical for NRC4 localization, oligomerization, and phospholipid association, underscoring its central role in resistosome formation and activation, but is not required for membrane localization during infection. We further show that this feature is conserved in NRCs and some singleton NLRs, but degenerated in NRC-dependent sensor NLRs, suggesting evolutionary structural specialization. Together, these findings provide new insights into the molecular basis of plant immune receptor function.
The first author, Hung-Yu Wang, conducted this work as a PhD student in the TIGP-MBAS program at Academia Sinica and is currently a postdoctoral researcher at Imperial College London.
A hydrophobic core in the coiled-coil domain is essential for NRC resistosome function