[Matzke, Antonius; Matzke, Marjori] A GFP splicing reporter in a coilin mutant background reveals links between alternative splicing, siRNAs and coilin function in Arabidopsis thaliana
POST: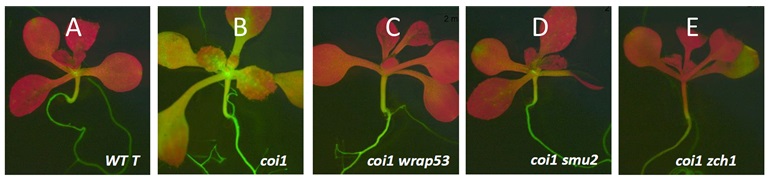
Figure legend: GFP phenotypes in coilin suppressor screen mutants (A, B) Intermediate GFP fluorescence in the WT T line compared to the hyper-GFP phenotype in coi1 single mutants. The coi1 suppressor screen identified mutations that attenuate the hyper-GFP phenotype of the single coi1 mutant: (C) coi1 wrap53; (D) coi1 smu; (E) coi1 zch1 double mutants.
Coilin is an intriguing protein that is present in most multicellular eukaryotes. Even after decades of research, the full range of coilin’s functions and modes of action are not fully understood. Coilin is best known as an essential structural constituent and prominent marker of Cajal bodies (CBs). CBs are nonmembranous nuclear organelles that are typically situated adjacent to the nucleolus, a second membrane-free nuclear organelle that also contains detectable amounts of coilin. CBs are major sites for maturation, processing, and quality control of small nuclear RNAs, including those that are present in small nuclear ribonucleoprotein particles (snRNPs) required for pre-mRNA splicing, and small nucleolar RNAs (snoRNAs), which guide processing and maturation of rRNAs and tRNAs in the nucleolus. To study coilin function in plants, the Matzke group carried out a genetic suppressor screen using a coilin (coi1) mutant in Arabidopsis thaliana and performed an immunoprecipitation-mass spectrometry analysis on coilin protein. The coi1 mutations modify alternative splicing of a GFP reporter gene, resulting in a hyper-GFP phenotype in young coi1 seedlings relative to the intermediate wild-type level. As shown in the study by Kanno et al., this hyper-GFP phenotype is extinguished in older coi1 seedlings by posttranscriptional gene silencing triggered by siRNAs derived from aberrant splice variants of GFP pre-mRNA. In the coi1 suppressor screen, identified suppressor mutations in WRAP53, a putative coilin-interacting protein; SMU2, a predicted splicing factor; and ZCH1, an incompletely characterized zinc finger protein. These suppressor mutations return the hyper-GFP fluorescence of young coi1 seedlings to the intermediate wild-type level. Additionally, coi1 zch1 mutants display more extensive GFP silencing and elevated levels of GFP siRNAs, suggesting the involvement of wild-type ZCH1 in siRNA biogenesis or stability. The immunoprecipitation-mass spectrometry analysis reinforced the roles of coilin in pre-mRNA splicing, nucleolar chromatin structure, and rRNA processing. The participation of coilin in these processes, at least some of which incorporate small RNAs, supports the hypothesis that coilin provides a chaperone for small RNA trafficking. Our study demonstrates the usefulness of the GFP splicing reporter for investigating alternative splicing, ribosome biogenesis, and siRNA-mediated silencing in the context of coilin function.