VgrG C-terminus Governs Transport Specificity of Type VI Secretion Effectors
Devanand Bondage, Jer-Sheng Lin, Lay-Sun Ma, Chih-Horng Kuo, and Erh-Min Lai
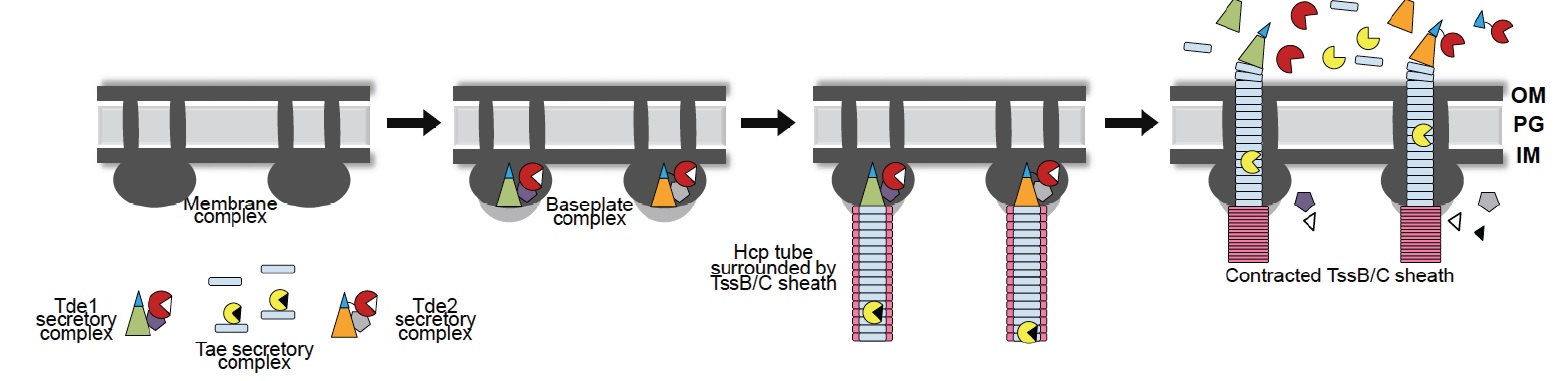
Type VI secretion system (T6SS) is a molecular weapon used by many Gram-negative bacteria to inject effectors into eukaryotic host cells or competing prokaryotes for bacterial survival and fitness. T6SS involves multiple strategies for effector delivery via fusion or interaction of effectors to structural components of the phage tail-like structure. However, the identity of the molecular components and mechanisms that govern the delivery of type VI effectors remain limited. The research team studied the delivery mechanism of antibacterial type VI secretion effectors, namely Tde1 and Tde2 DNase toxins, in Agrobacterium tumefaciens. They identified the protein components including the spike protein (VgrG1 and VgrG2), adaptor/chaperone, and a sharp tip PAAR protein are required for delivery of cognate Tde toxin. Importantly, the divergent C-terminus of VgrG1 and VgrG2 spike proteins governs the Tde toxin delivery specificity. In-depth genetic and biochemical evidence demonstrated that VgrG1 C-terminus is the molecular determinant required for interacting with a specific adaptor/chaperone protein and PAAR tip protein to govern Tde1 translocation across bacterial membranes and delivery into target cells for antibacterial activity. Further genomewide survey discovered the widespread presence of these genetic modules in various Proteobacteria, suggesting a conserved mechanism in type VI effector delivery.
Proceedings of the National Academy of Sciences USA 113 (2016): E3931-40.
DOI: 10.1073/pnas.1600428113